The Architect of Regenerative Medicine
Defining the Science of Calcium Hydroxylapatite
Welcome to MyCaHA.com, the central intelligence hub for Calcium Hydroxylapatite (CaHA) technology. We bridge the gap between material science and clinical practice, providing evidence-based analysis on the world’s most versatile bioceramic. Whether you are investigating the rheological properties of dermal fillers, the osseointegration of dental implants, or the manufacturing standards of microspheres, you have arrived at the source.
THE MOLECULE
Stoichiometry & Biocompatibility
Calcium Hydroxylapatite is not merely a filler; it is a biomimetic scaffold. Synthetically produced to be chemically and structurally identical to the primary mineral constituent of human bone and teeth, CaHA offers a unique safety profile characterized by inherent biocompatibility and non-toxicity
The CaHA Distinction:
Chemical Precision: Defined by the formula
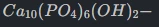 . Medical-grade CaHA maintains a calcium-to-phosphorus (Ca/P) molar ratio of approximately 1.67, mirroring endogenous bone mineral.
. Medical-grade CaHA maintains a calcium-to-phosphorus (Ca/P) molar ratio of approximately 1.67, mirroring endogenous bone mineral.Microsphere Engineering: For soft tissue applications, raw CaHA is sintered at high temperatures (>1000°C) to create smooth, non-porous microspheres. This specific morphology is critical; it allows the particles to serve as a scaffold for fibroblast attachment without eliciting a chronic foreign body granulomatous reaction.
Metabolic Pathway: Unlike permanent synthetic polymers, CaHA is biodegradable. It follows a natural metabolic pathway, degrading into calcium and phosphate ions which are eliminated via the renal system, leaving behind only the patient's newly generated tissue network.
MECHANISM OF ACTION
Immediate Volumization
Upon injection, the aqueous gel carrier (typically Sodium Carboxymethylcellulose) provides an immediate 1:1 correction of the defect, resisting compressive forces due to high Elastic Modulus ($G'$) and viscosity
The Scaffold Effect
As the carrier gel is resorbed over 2–3 months, the CaHA microspheres (25–45 µm) remain in situ. They create a thixotropic lattice that supports fibroblastic ingrowth.
Neocollagenesis & Elastogenesis
The physical tension and surface interaction of the microspheres stimulate fibroblasts to synthesize new Extracellular Matrix (ECM) components. Clinical histology confirms a transition from Type III (reticular) collagen to Type I (structural) collagen, alongside significant increases in elastin and proteoglycans.
CLINICAL APPLICATIONS
Aesthetic Medicine & Dermatology
CaHA is the gold standard for structural facial rejuvenation and hand augmentation. Modern techniques utilize "hyperdiluted" CaHA washes to improve skin quality and laxity in the neck, décolletage, and body by maximizing surface area contact with fibroblasts.
Other Products: Radiesse® (Merz Aesthetics), HArmonyCa™ (Allergan), Renú® (Cytophil)

Laryngology & Speech Patholog
In the treatment of vocal fold insufficiency and paralysis, CaHA acts as a durable bulking agent. Injected into the vocal folds, it restores phonation and glottic competence, offering a long-lasting alternative to temporary resorption materials
Other Products: Renú® Voice, Radiesse® Voice
Urology
For female stress urinary incontinence (SUI), larger particle CaHA (75–125 µm) is utilized as a bulking agent to reinforce the urethral sphincter. The particle size is specifically engineered to prevent migration while providing durable occlusion
Other Products: Coaptite®
Orthopedics & Dentistry
CaHA coatings on titanium implants accelerate osseointegration, creating a chemical bond between the implant and the patient's bone. Additionally, synthetic CaHA granules serve as effective bone void fillers for trauma and spinal fusion
REGULATORY & MANUFACTURING STANDARDS
Precision Engineering
The production of medical-grade CaHA requires adherence to rigorous international standards to ensure safety and efficacy.
Standards
Material specifications are governed by ASTM F1185 (Standard Specification for Composition of Hydroxylapatite for Surgical Implants) and ISO 13779
Quality Control
Manufacturers utilize X-Ray Diffraction (XRD) to verify crystallinity (>95%) and ICP-MS to ensure heavy metal limits are far below safety thresholds
Regulatory Status
CaHA fillers are generally regulated as Class III Medical Devices by the FDA, requiring Premarket Approval (PMA) or 510(k) clearance demonstrating substantial equivalence
Partners and references
Use this section to highlight our commitment to healthcare innovation.

Clinics
Care4Me Clinics

Distributors
Looking for Regional Partners

Technology
Sabhya Technologies, Malkan Solutions